A Case of Deja VuThe recent spate of articles about buckminsterfullerene (Bf), named for "the inventor of the geodesic dome," is reminiscent of what happened in the 1960s when x-ray crystallographers Donald Casper and Aron Klug discovered the icosahedral shape of many of the viruses. "The protein outside the coat, far from being a 'bag,' is a beautifully constructed geometrical figure... many of which are like the architecturally famous domes of Buckminster Fuller" wrote Dean Fraser in his Viruses and Molecular Biology (Macmillan, 1967). Such references were typical. As Fuller recounts in Utopia or Oblivion (p. 104): "The virologist discovered the proliferation of my geodesic structures, which had emerged from structuring aspects of my energetic geometry studies..." |
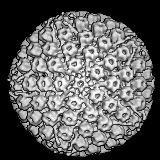 |
| The deeper connection between virology and synergetics is only hinted at by this micro-macro structural analogy however. For Fuller, the domes were simply artifacts that had "emerged" from his geometric investigations. "In 1957 Klug asked if I could identify the geodesiclike protein shell of the polio virus. I was able to give him the mathematical explanation of the structuring." |  |
| In Critical Path, Fuller records his contribution in the appended chronology under the year 1963: "World Congress of Virologists meeting Cold Spring Harbor, Long Island, N.Y., announce comprehensive discovery of protein shells of viruses, which they publicly acknowledge (featured, front page, New York Herald Tribune) as having been anticipated by RBF's formula of frequency to the second power of 10 plus 2." This expression 10F2+2 is what Fuller intended for use as a bridge to synergetics. Those who traced this formula from its front page mention in the newspaper to its origin in synergetics would come across its more general form: 2PF2+2, an expression at the heart of Fuller's investigations into sphere packings. |  |
| By setting P=5
in the expression In a virus, the
RNA-protecting shell or capsid is made from sub-units
called capsomeres. By taking F as the number of
between-capsomere intervals, and using |
 |
| X-ray diffraction patterns showed these viruses to have icosahedral symmetry. How does the sphere packing formula derived for the cuboctahedron manage to predict the number of spheres in an icosahedrally shaped capsid? Again, Fuller hoped that his audience of virologists would be led by this question to his jitterbug transformation. The jitterbug motion, a combination of rotation and contraction, smoothly transforms the cuboctahedron into an icosahedron and vice versa. More generally, the jitterbug transformation serves as a unifying motion relating 4- and 5-fold symmetric polyhedra. |  |
| When applied
to shells of closest packed spheres, the jitterbug
transformation turns the cuboctahedral shell into an
inherently non-nucleated one. The icosahedral arrangement
is essentially hollow. It does not have room for
concentric shells to be formed by closest packing -
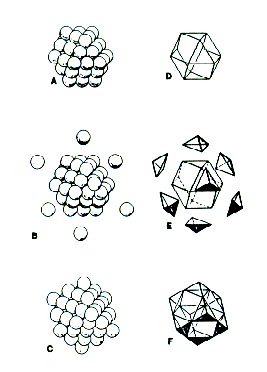
unless we allow the spheres to deform. Chemists have been able to generate icosahedral clusters of atoms by forcing xenon gas through a nozzle at high pressure. The xenon clusters, which spontaneously form as the gas cools, consist of concentric icosahedral shells - named Mackay shells after crystallographer Alan Mackay. The total number of atoms in such clusters is expressed using the familiar synergetics formula: N = 1 +SIGMA(10F2+ 2) Or, alternatively, one may derive:[2] N = (10/3)F3+ 5F2+(11/3)F + 1 This icosahedral cluster growth pattern cannot be maintained indefinitely out to any frequency however. At some point, it jitterbugs to release pent-up stress and relaxes into alignment with the isomatrix to become a stable crystal. As Dr. Bjornholm of the Niels Bohr Institute at the University of Copenhagen puts it: "The point is that hard spheres cannot really be packed into the icosahedron... The spheres have to be soft... As a result a strain builds up in the cluster and at some stage it becomes favourable to release the strain through a rearrangement of the atoms leading to the familiar cuboctahedral structure of the macroscopic crystal."[3] The jitterbug transformation has been discovered in nature. Spheres may be closest packed concentrically and omnisymmetrically to form shapes other than the cuboctahedron. Fuller discovered that by changing the value of P in his expression 2PF2+2, he was able to accommodate sphere packings which conformed to the external contours of other "primary systems." A tetrahedral packing is generated when P=l, an octahedral one when P=2, and a cubic packing when P=3. |
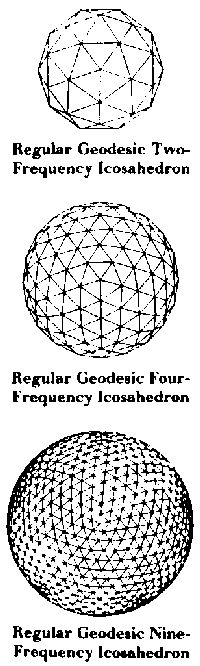 |
| When P=2, for
example, the expression becomes 4F2+2, and
gives the sphere populations in an octahedron's
successive layers. Six spheres form an octahedron
centered around a void while 18 build around a nuclear
sphere to form a 2-frequency octahedron and so on. 6F2+2 describes the two packing arrangements which define a cube of any frequency: the body-centered cubic (bcp) and the one generated by tangent spheres inscribed in XYZ cubes.[4] These packings are not necessarily nucleated however (the tetrahedron acquires its first nucleus at frequency 4), nor is the cube as densely packed as the isomatrix will allow at some frequencies. The formation of nucleated densest packings of various shapes, excluding the inherently non-nucleated 5-fold symmetric polyhedra, is another, related topic which Fuller investigates in Synergetics and Synergetics 2 .[5] Fuller hoped that these investigations would prove useful in chemistry, where the arrangement of protons and neutrons in atomic nuclei might be modeled by sphere packings of various shapes. |
 |
| Contrary to Fuller's hopes, however, 10F2+2 did not go down in history as a special case of 2PF2+2. It was instead subsumed by another expression. Casper and Klug found that Fuller's 10F2+2 did not account for all of the capsomere counts they were getting. They needed to accommodate the so-called "skew" cases corresponding to what later came to be known as Class III geodesic structures. In such cases, the rows of capsomeres are not parallel to the edges of an icosahedron but cross them at an angle. Their new expression was 10T2+2, where T=b2+bc+c2. Casper and Klug's expression reduces to Fuller's when c=0. |  |
| We can suppose that Fuller's input helped Casper and Klug to derive their expression but, once published, it was incorporated into the literature in a way that did not link to Fuller. In his Virus Macromolecules and Geodesic Domes (1967), for example, H.S.M. Coxeter traced the newly emerging classification scheme for geodesic structures to mathematician Michael Goldberg's paper, A Class of Multi-Symmetric Polyhedra which appeared in 1937 (Tohoku Mathematics Journal, 43, 104 -108). | |
| Coxeter rather
patronizingly implied that Fuller-the-architect might
learn something from a real mathematician:
"Summarizing our conclusions, we may say that
Goldberg's classification of 'multi-symmetric' polyhedra
yields a convenient classification of possible shapes for
virus macro-molecules and geodesic domes. The possibility
b <> c may perhaps inspire Fuller to make new domes
which, like pineapples and helices, have no planes of
symmetry."[6] The fact that |
 |
| Synergetics
was most likely ignored during this period because it was
as yet only available in fragments. By the mid-1980s,
when articles about buckminsterfullerene began to
proliferate, the situation had changed: Synergetics and
Synergetics 2 had appeared, in 1975 and 1979
respectively. Now scientists could discover what Fuller
was up to in his "energetic geometry studies"
as they could not so easily during the virus scenario. The synergetics formula 10F2+2 may be
modified to |
 |
| In all of
these structures, however, the number of pentagons is
always 12, a fact which intrigued the virologists:
"It should be noticed that the subunits are not
identically located: most of them are surrounded by six
others, but those at the corners of the icosahedron have
five neighbors. Recent work shows that the protein
subunits at these vertices of adenovirus are both
serologically and morphologically different from the
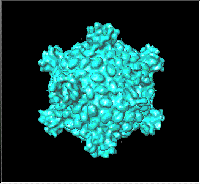
rest." (Fraser, 1967, pg. 53). This same spherical pattern of 12 pentagons and a variable number of hexagons occurs in another biological structure known as a "coated vesicle," which has an external lattice analogous to the viral capsid, known as clathrin. As mathematician Ian Stewart points out in Circularly covering clatharin (Nature, Vol 351, 9 May '91, pg. 103), the hexapent cage illustrates the optimal solution to the problem of how to cover a sphere with the least number of partially overlapping circles of the same radius. Mathematicians had hitherto missed this optimum fullerene-like solution. "Not for the first time, nature has beaten them at their own game" writes Stewart. |
 |
| Sections: 1 2 3 4 5 6 7 | |